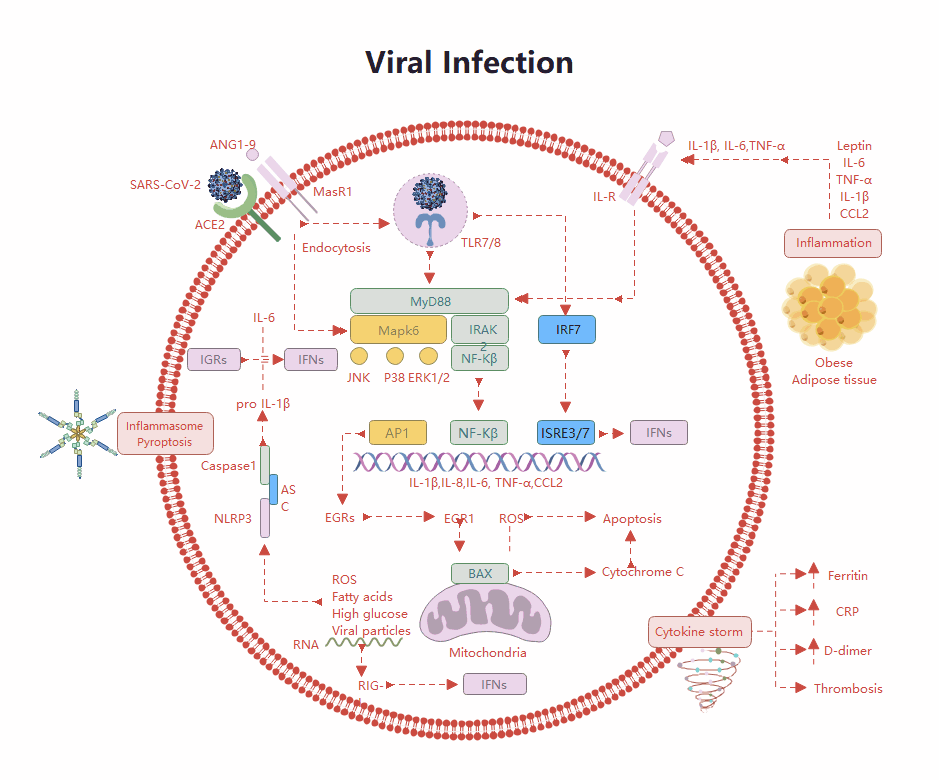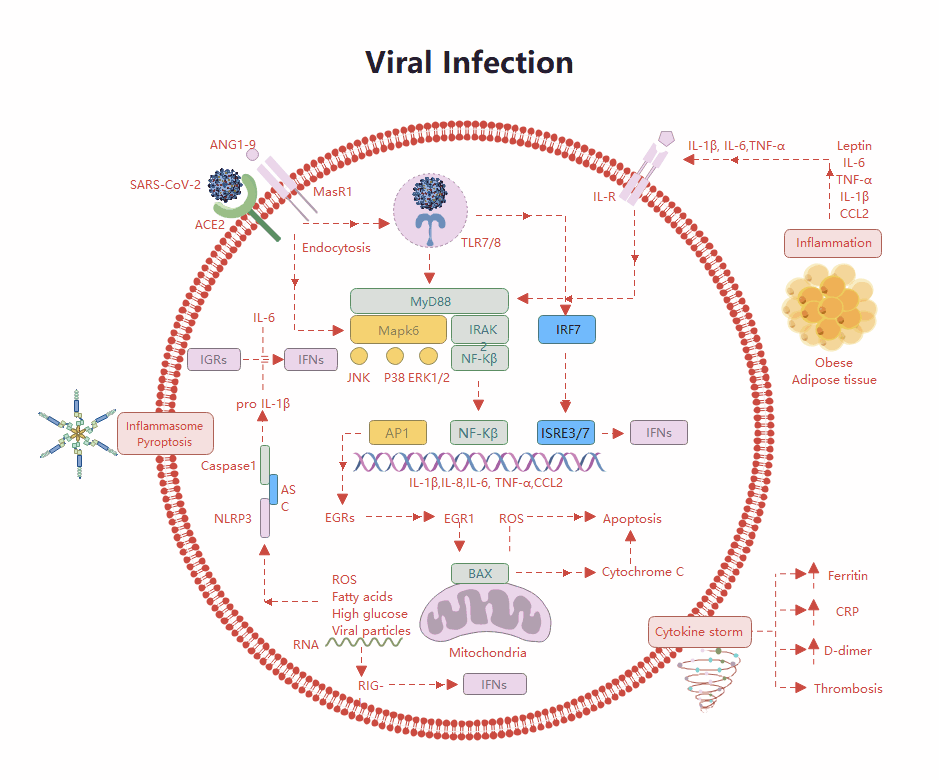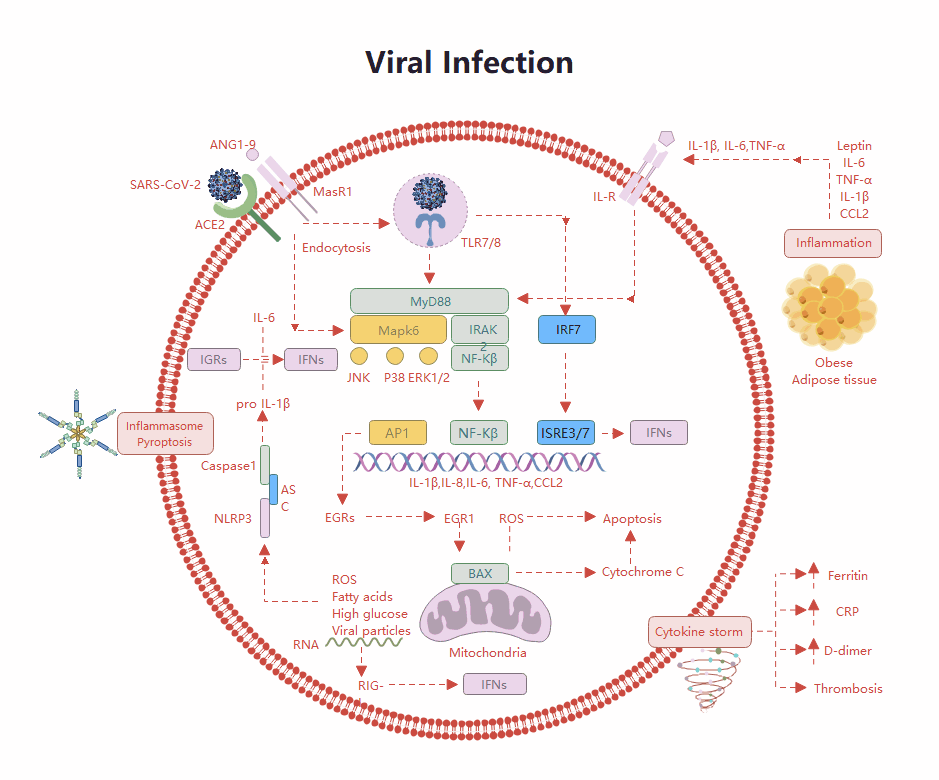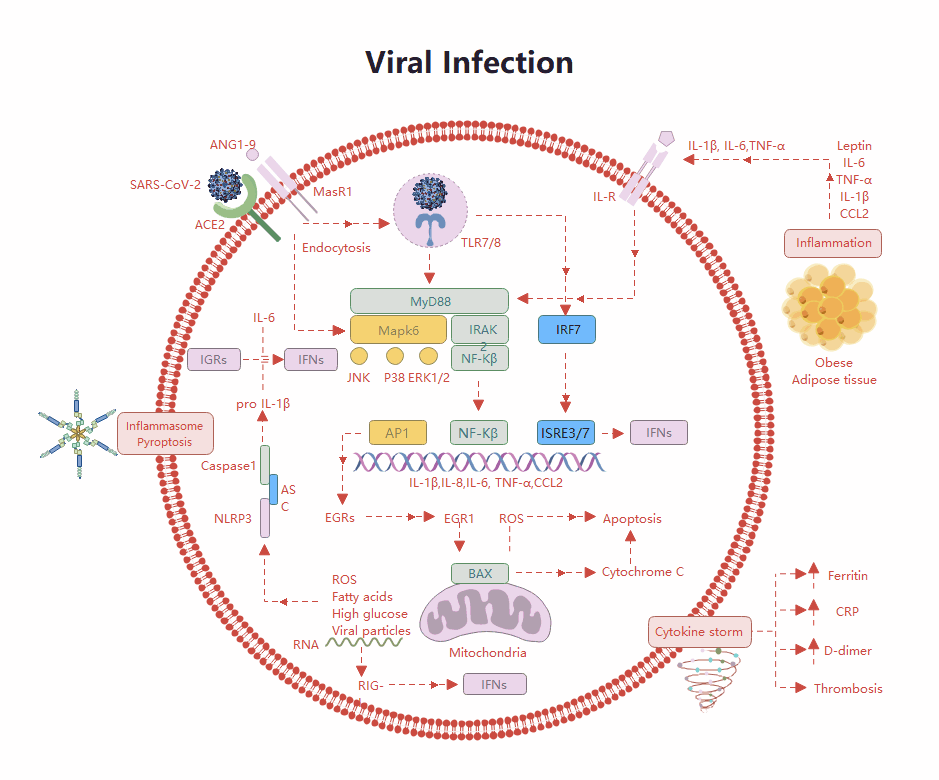
Templates Community
Discover, learn and get inspired by 0+ free editable templates for your next project!
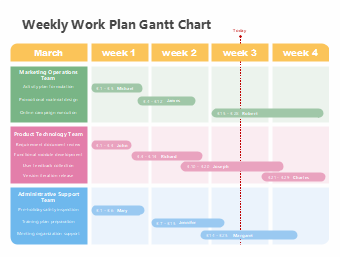
207
33
0

149
13
1

119
26
1

135
23
3
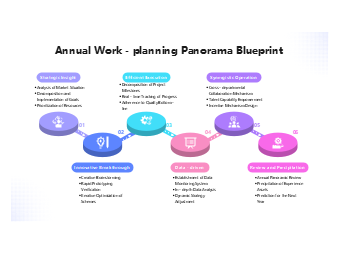
138
26
0
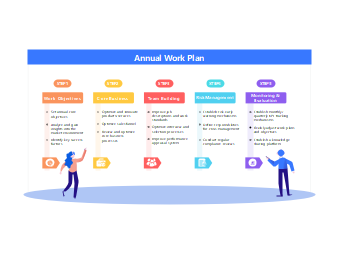
111
27
3
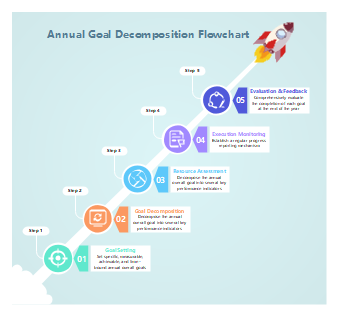
97
24
0
152
55
0
75
5
1
65
2
0