
Templates Community /
Top 15 Oncology Targets-Phase Distribution Pipeline Table
Top 15 Oncology Targets-Phase Distribution Pipeline Table
WSIrbAxv
Published on 2024-01-06

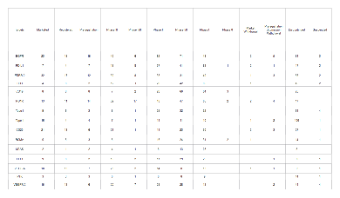
This Top 15 Oncology Targets Phase Distribution Pipeline Table systematically presents the clinical phase distribution and development status of drugs targeting 15 high-impact oncology proteins, including key targets like EGFR, PD-L1, ERBB2, CD3, CD19, PD1R, and VEGFR-A. Tailored for the biopharmaceutical, clinical research, and oncology industries, it serves as a critical reference for drug development scientists, clinical researchers, pharmaceutical portfolio managers, and market analysts focused on oncology therapeutics.
The table categorizes each target’s drug candidates by full lifecycle stages: Marketed, Registered, Preregistration, Phase 0, Phase I, Phase II, Phase III, Phase II/III, Phase III/IV, Market Withdrawal, Preregistration Submission Withdrawal, Discontinued, and Suspended, with precise numerical counts for every category. It empowers stakeholders to quickly assess the competitive landscape, track clinical progress across development phases, and analyze attrition rates for oncology drug candidates. This data-driven tool supports strategic R&D planning, portfolio optimization, competitive benchmarking, and market trend forecasting, enabling data-informed decision-making for drug development programs.
Fully editable, this template allows teams to update target lists, refine stage categorizations, and refresh numerical data as new clinical trial results, regulatory updates, or market withdrawals emerge, ensuring the pipeline analysis remains current and actionable.
Tag
Pharmaceutical R&D Management
Drug Development Pipeline
Oncology Targets
Share
Report
0
15

Post
Recommended Templates
Loading

 Desktop
Desktop