
Template Community /
NaF_PID
NaF_PID
James Ruff
Published on 2021-11-29

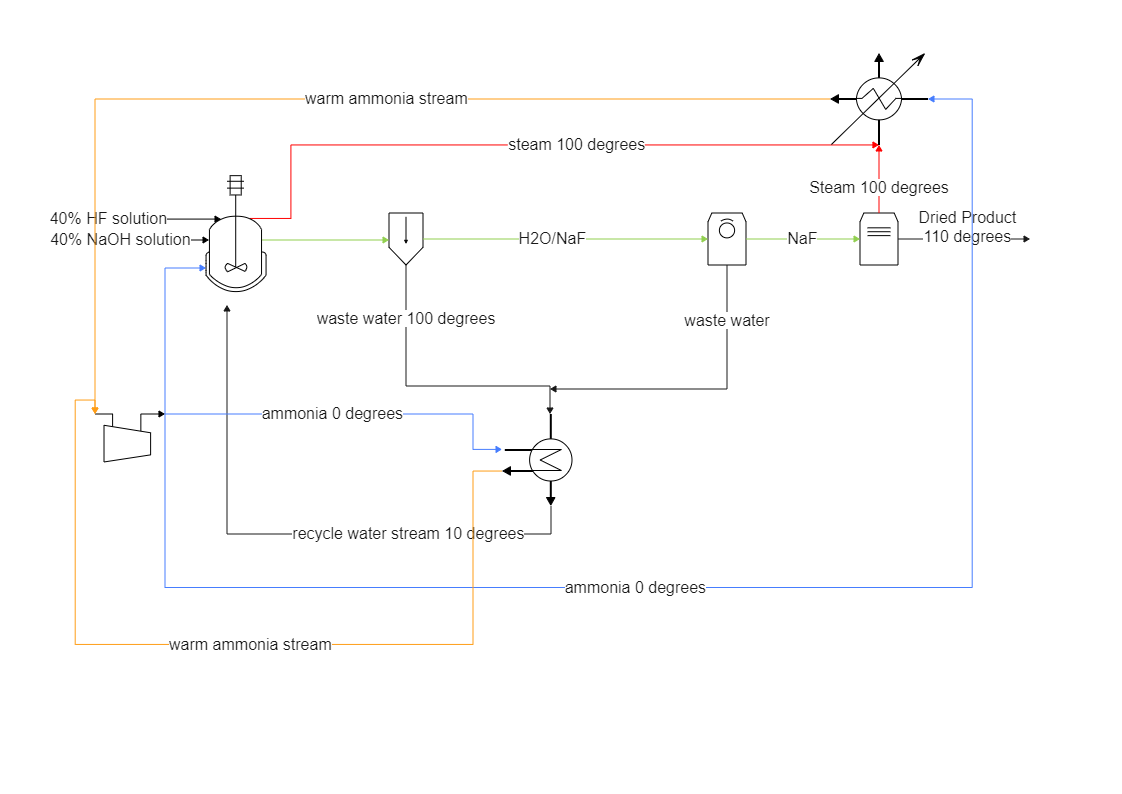
Sodium Floride (NaF) PID Diagram shows how solid sodium fluoride is dissolved into water; it completely dissociates into sodium and fluoride ions. P&ID is the acronym for "Piping and instrumentation diagram," i.e., a very detailed diagram showing the processes happening within a plant, the involved equipment, and their interconnections. As the P&ID diagram illustrates, the sodium ions do not react with water, but a minor portion of the fluoride ions will deprotonate water, producing small amounts of hydrofluoric acid and hydroxide ions. In simpler words, the reactant (sodium chloride, or NaCl) is different from the products (sodium cation and chloride anion).
Tag
Building Plan
diagram
Share
Report
1
106

Post
Recommended Templates
Loading